
Source: hofmannlab.msi.ucsb.edu
BLOG: Is the oceans’ power to maintain life rivaled by our own power to destroy it?
By Ashley Westgate MSEP ’15 and Keston Finch MSCSP ’15
In the wake of the recent IPCC AR5 report, scientists have highlighted the added stress that increased anthropogenic CO2 is placing on our world’s ocean systems. Oceans are becoming increasingly acidic due to increased carbon dioxide absorption and other land based sources of pollution, which, if left unchecked, could destroy all coral reefs, disrupt ocean systems and entire food chains, and threaten available fishery resources. Dr. Gretchen Hofmann, marine ecophysiologist, and professor of marine biology and ecology at UC Santa Barbara, spoke on this issue at the Bard Center for Environmental Policy’s National Climate Seminar this week, where she argued for addressing the cause and also dealing with the immediate problem. By explaining some of her own research on marine organism responses to ocean acidification (OA), she demonstrates how the unification of environmental networks can help expand existing scientific knowledge which can then be used as framework for stimulating new policy.
Over the last few decades, the topic of climate change has concentrated immensely on intense temperature fluctuations which contributes to the loss of sea ice within polar regions, accelerated sea level rise, which threatens island nations and coastal zones and the increase voracity of storms and other natural disasters. But it is not until recently have scientists recognized one of the most serious outcomes of such increased anthropogenic CO2: ocean acidification.
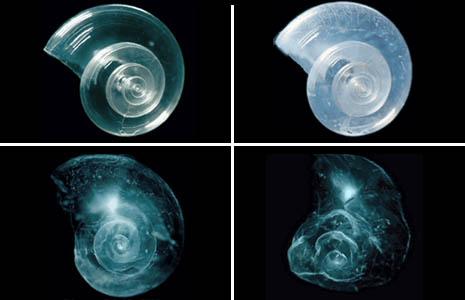
Source: http://ocean.si.edu/ocean-photos/shell-dissolves-seawater
Dr. Hofmann, whose research centers on marine organism responses to OA, addresses this question. She explains that while the issue of climate change can and has been debated there is no denying the simple chemistry of CO2 in seawater which, when absorbed, forms carbonic acids which lowers the natural pH required for biological functioning. While scientists have made great strides in the past four years on understanding OA, there is still a significant amount of work to be done in determining where decreasing pH levels participate in triggering molecular responses in organisms. For example, the physiological response of coral reefs to decreased pH results in bleaching, but this result is different in organisms within estuaries or fish communities, and these all will differ by region. The effects of multiple stressors on a system, like temperature increases, pollution and oxygen depletions, can also compound acidification effects.

Source: http://www.epa.gov/climatestudents/impacts/signs/acidity.html
Hoffman spoke broadly on ecosystem impacts, evolutionary adaptation and new technologies used in understanding carbonate chemistry. She also stressed that biological impacts from ocean acidification would result in a change in existing synergies, for example predator/prey relationships, leading to the extinction of important primary ocean producers and other significant ecoystemic shifts. This is not to say that our world will not survive because of rising CO2 levels, but the environment overall may transform into something very different than what we are used to.
Empirical evidence on the failing pacific north-west oyster industry show that this issue is slowly forcing farmers to move their operations to other regions like Hawaii because of concerns over OA’s impacts on hatchery production. These subtle attacks on food security and regional economies are now forcing us as a people to ask: is the oceans’ power to maintain life rivaled by our own power to destroy it?
New technologies, however, have enabled researchers to make more robust scientific predictions of the effects of OA compared to their previous attempts. Our ability to record ocean pH, for example, has been revitalized by the development of new technologies like the CFET biosensor. This sensor is allowing scientists to continually record ocean pH, advancing our understanding of ecosystem tolerances. Recent recordings show upwelling on Californian coral reefs that brings CO2-rich water into the ecosystem. With this established, scientists can observe how the coral reefs respond and also initiate predictions of what ocean acidification will do to this region.
The Way Forward
Dr. Hofmann, in her closing remarks, stated “community based knowledge to action at the state level are where we are able to be most productive in addressing OA”. The Washington Blue Ribbon Panel, California Current Acidification Network (CCAN), the West Coast Ocean Acidification and Hypoxia Science Panel (OAH), the West Coast Governors Alliance on Ocean Health (WCGA), numerous other community groups, and national partners have banned together, in various ways and venues, to help create legislation, promote activities and strengthen their research capacity in understanding and dealing with the effects of OA within their locally significant marine ecosystems. Furthermore, these collaborative processes also call for us to adopt more sustainable practices and ocean conservation strategies which would reduce the effect of other combined stressors – such as pollution, overfishing and habitat destruction which would allow marine ecosystems to be resilient enough to combat this issue of ocean acidification. If not, we might as well start working on the world’s largest Alka Seltzer® tablet.
Join the National Climate Seminar live, it takes place via conference call the first and third Wednesday of each month during the academic year. Connect with top scientists, analysts, and political leaders to talk about climate change.
To listen to this and other National Climate Seminars, click here.
Blog student Authors:



